Meridian Medical Technologies, a Pfizer company and Mylan’s® manufacturing partner for EPIPEN® (epinephrine injection, USP) Auto-Injector, has expanded a voluntary recall of select lots of EPIPEN and EPIPEN JR® (epinephrine injection, USP) Auto-Injectors to now include additional lots distributed in the U.S. and other markets in consultation with the U.S. Food and Drug Administration (FDA).
BioRidge, Mylan’s® third-party administrator of the EPIPEN4SCHOOLS® program, proactively tracks the lot numbers of the product distributed to all schools enrolled in the program.
The schools that have EPIPEN and/or EPIPEN JR Auto-Injectors affected by the recall have been identified and:
- Have been notified by email and first-class mail
- Will receive replacement product via an automatic shipment from BioRidge
- Will receive a package containing instructions on how to return the affected auto-injectors in a prepaid mailer
Schools do not need to take any action to initiate this process.
Schools that we have identified as NOT having received product subject of the recall will receive an email indicating such. Out of an abundance of caution, those schools will be instructed to check the lot numbers of the EPIPEN Auto-Injectors in the chart provided below. If the devices are included in the recalled lots, please call BioRidge immediately at 973-845-7600.
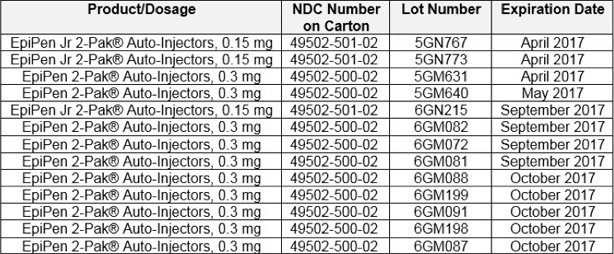
The recall impacts the below listed lots of the 0.3 mg and 0.15 mg strengths of EPIPEN Auto-Injector:
The lot number is located on both the 2-Pak carton and the auto-injector. If the recalled EPIPEN Auto-Injector is still contained in the carton, refer to the left flap on the carton, which is black in color. You will find the lot number written in white and preceded by the word ‘LOT.’
On the auto-injector itself, you will find the lot number towards the top of the label in black and preceded by the word ‘LOT.’
NOTE: The NDC on the box ends with “2” because it contains two EPIPEN Auto-injectors. The NDC on the individual EpiPen within the box has an NDC ending in “1.”

For more details on the recall, please see mylan.com/epipenrecall and the press release on mylan.com.
If students have personal EPIPEN® Auto-Injectors that are kept at the school, we ask that patients and caregivers are informed. Patients with personal EPIPEN Auto-Injectors affected by the recall follow a different return and replacement process. Patients and caregivers should visit http://www.mylan.com/epipenrecall for instructions on how to identify if their auto-injector is part of the recall and initiate a request for a replacement if affected. Mylan is committed to replacing recalled devices at no cost.
If you have questions or concerns about the product in your school please call BioRidge at 973-845-7600. If you had additional questions about the recall, please call Mylan Customer Relations at 800-796-9526.